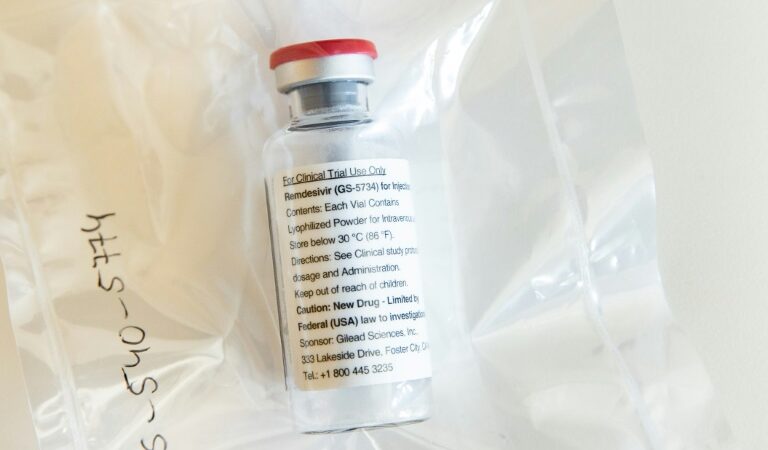
The US Food and Drug Administration (FDA) approved on Friday an experimental antiviral drug, Remdesivir, for emergency use in COVID-19 patients. The drug can now be given to patients who are hospitalised with severe symptoms and need supplemental oxygen therapy or a ventilator. Emergency use authorisation means that unapproved drugs that might help treat a life-threatening disease can be used when no approved alternatives are available.
Investigators probing deaths of six people who contracted vaping-related lung diseases have warned that NO e-cigarette is safe. Despite vaping industry officials trying to blame illegal cannabis products, the US Centres for Disease Control and Prevention (CDC), said people should stop using any electronic cigarette or vaping device. The FDA has warned consumers against using vaping devices bought “on the street”.