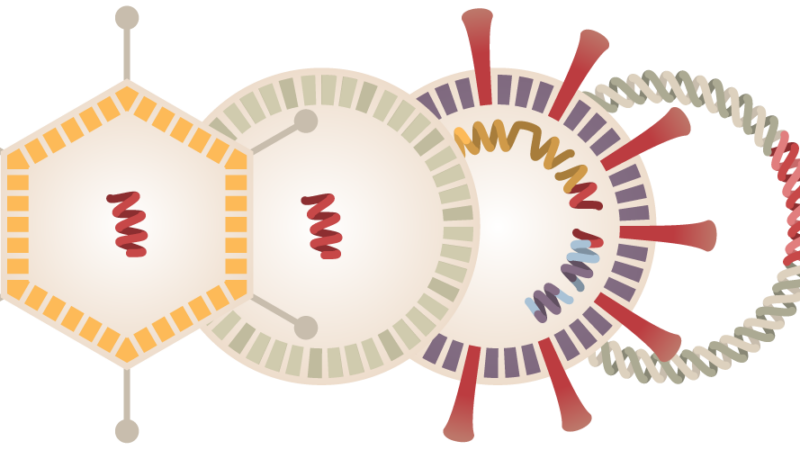
A Chinese-made vaccine for coronavirus has entered the final stage of testing in Brazil. The vaccine, developed by private Chinese pharmaceutical firm Sinovac Biotech entered the final phase after volunteers received the first doses of the drug. It became the third in the world to enter Phase 3 clinical trials, or large-scale testing on humans — the last step before regulatory approval.
A human trial of a potential coronavirus vaccine is being launched by Oxford University with the aim of making it available to the public. In its first phase, half of 1,112 volunteers will receive the potential vaccine against COVID-19, the other half a control vaccine to test its safety and efficacy. The volunteers are aged between 18 and 55, are in good health.